Skin Wars: Prevent Skin Infections | Fight Skin Fungus Naturally
Skin Bacteria: Fighting the War of Skin Infections and Skin Fungus
“The mutualistic bacteria within your skin flora are fighting for your skin's protection. There are ever-present pathogenic bacteria that are waiting for their opportunity to strike. Keep your forces strong by maintaining the proper pH-balance for you skin.”

Support Your Troops and Fight Skin Infections and Skin Fungus Naturally
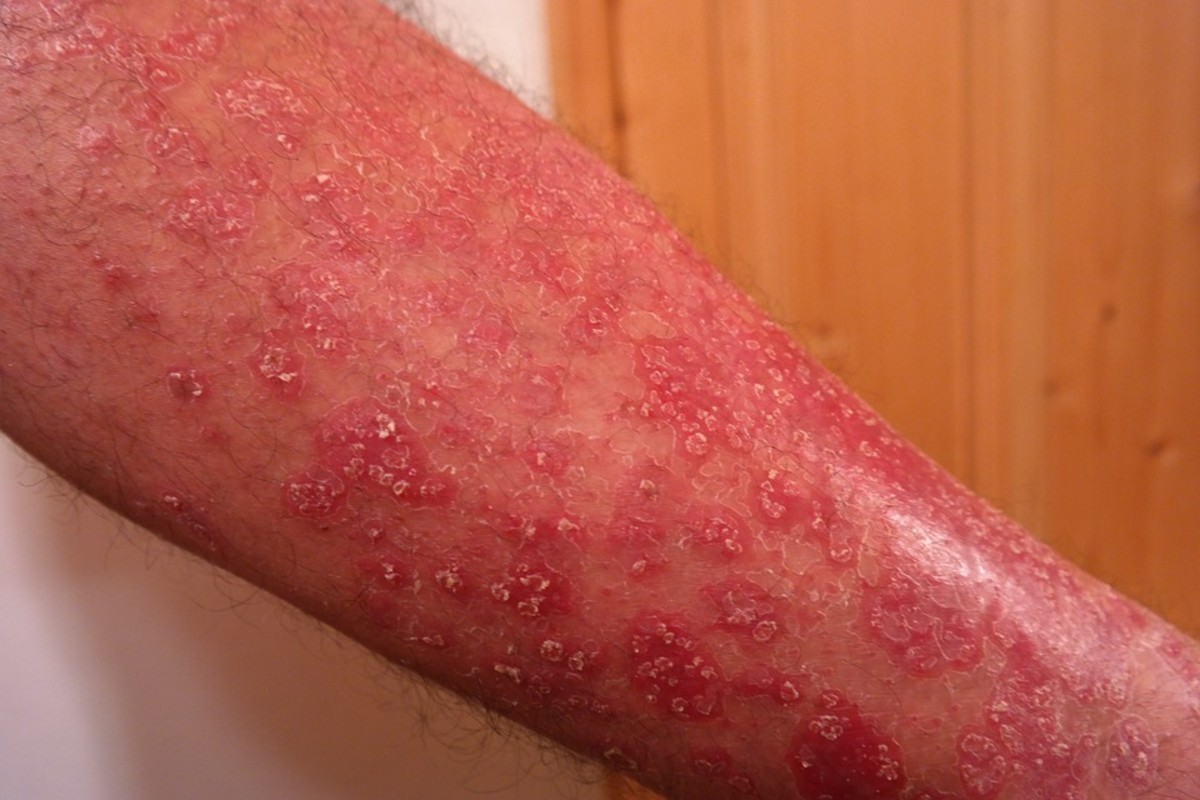
Your skin has 1 Trillion bacteria in constant battle. The war for supremacy between good bacteria and harmful pathogens determines whether or not you get skin infections, skin fungus or other skin related issues.
- As commander in chief of your skin's special forces (skin flora) it is your responsibility to support your troops so they can win the war against skin fungus and skin infections.
- Discover why your skin need the proper pH levels your skin needs to give the good bacteria the tactical advantage over the pathogens.
- Find out how you can keep your skin's pH at the optimum levels to defend against the ever present pathogens on your skin.
- Learn how scrubbing your skin with soap and hot water can lead to skin infections and skin fungus.
- Use a natural and gentle exfoliate to help keep your skin healthy without destroying all of your valuable skin flora.

Choose Your Battleground: The importance of your skin's pH level.
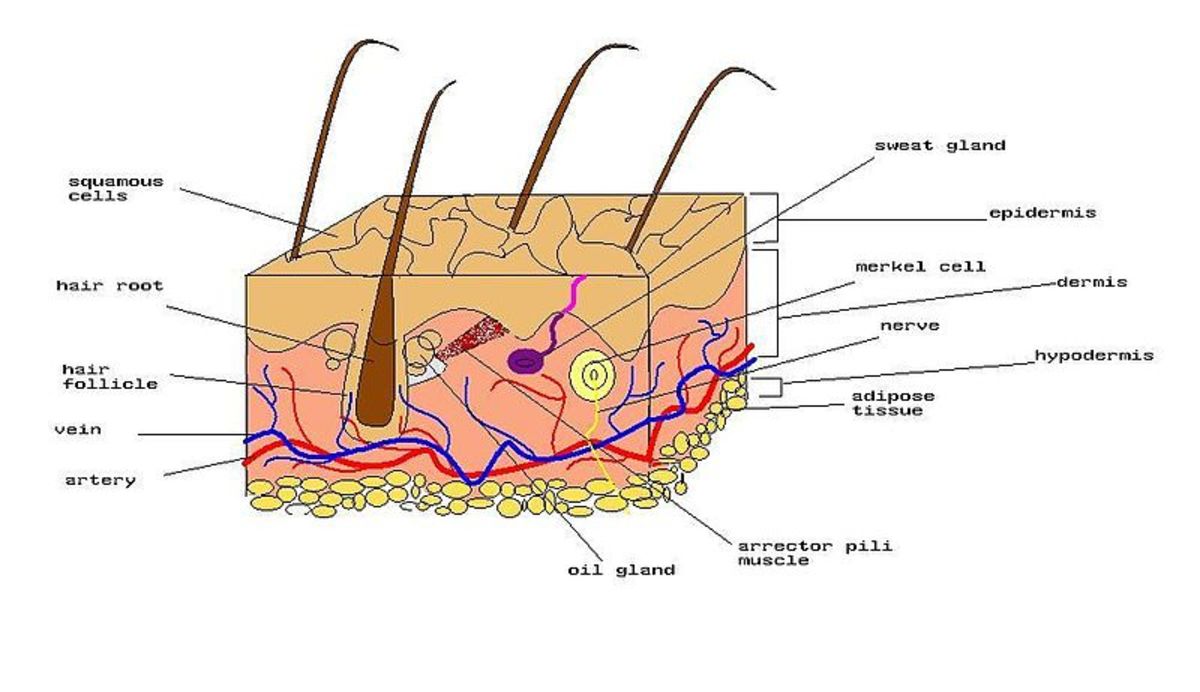
Your skin is a protective barrier, composed of dead skin cells, fatty acids, and lipids. It works to protect your body by creating acidic environment, at a pH between 4.4 and 5.5. Your skin forms what is called an "acid mantle".
The pH levels of your skin can support or hinder the effectiveness of beneficial bacteria. The acidic layer helps your skin retain moisture and keeps germs out. Your skin flora thrives in an acidic environment.
The pathogens that lead to skin infections and skin fungus don't do well in this environment. Think of it like positioning your skin flora on higher ground. While your skin flora fights from a dry vantage point, the pathogens are forced to fight from swampy waters.
On the flip side, if the pH rises too high, the pathogens have a better chance to break through your natural defense. When these pathogens get past the outer layer of protection you become susceptible to all kinds of skin ailments including infections and fungus. It also can trigger skin diseases like psoriasis or eczema.
How to Protect Your Skin's pH Levels
As with most things, prevention is the best cure. Protecting your skin's acid mantle is a preventative measure to help fight skin infections and skin fungus. Healthy, properly balanced skin is far less likely to get infections or trigger other skin conditions.
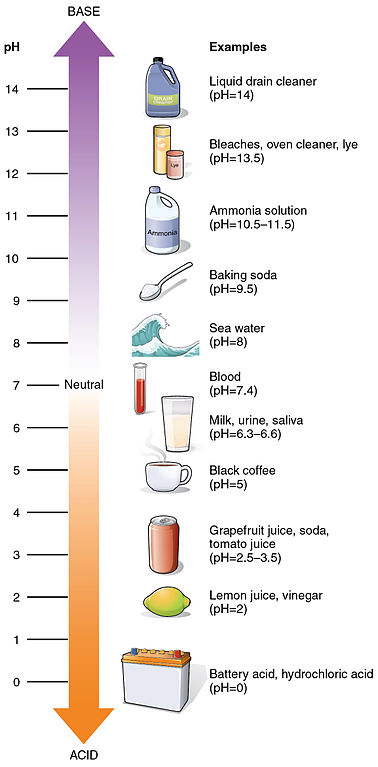
As you already know, pH is measured on a scale of 1-14. Distilled water is a 7. Everything below is acidic. Everything above 7 is alkaline.
If you had something that was very acidic and you wanted to neutralize it (bring it closer to 7) you want to add a base (alkaline) to the acid. In the case of your skin, you are trying to protect the acid layer, so you want to add an acid. Ideally something equal to or just slightly more acidic than the desired pH.
My go to acid mantle supporter is apple cider vinegar. It has a pH around 3. When it is mixed with water it is a little closer to 4, so it is very close to the desired pH your skin flora thrive in.
The Facts About Soap
-
Highly caustic, lye is 13.5 on the pH scale.
-
Lye is the catalyst that turns fats into soap.
-
Bar soap is made with sodium hydroxide lye.
-
Liquid Soap is made with potassium hydroxide lye.
-
Both soaps can raise the pH of your skin a point or more in on use.
-
It can take up to a day to recover your acid mantle from using soap.
Can Soap Lead to Skin Disease? Sort of...
On the pH scale we just referred to, vinegar resides in the lower of the acid range. The extreme reference for an acid is battery acid. There is obviously quite a difference between the acidity of battery acid and vinegar.
Apple cider vinegar is slightly less acidic than distilled vinegar. bringing it closer to the 3 range. Mixed with water it is closer to 4.
On the extreme alkaline side of the pH scale is lye. Only half a point off from drain cleaner. The highly caustic lye is the necessary ingredient to chemically change fats into soaps.
There are two types of lye sodium hydroxide which is used to make bar soap and potassium hydroxide which is use to make liquid soap. The two can not be interchanged.
Bar soap is by far the worst soap for your skin. With a typical pH level of 9 to 12 it is on the opposite side of the scale from your skin's ideal pH of 4.5-5.5.
A single washing with bar soap can raise your pH level a whole point or more. At this change it can take several hours, up to a day, for your skin to balance out.
While your skin works to balance itself out, your battalion of beneficial bacteria is swarmed by pathogenic bacteria taking advantage of the drop in defences.
In addition, the lye absorbs into your skin. Bar soap is made of sodium hydroxide, so your body is absorbing sodium. Most of us meet or exceed our daily allowance of sodium through our diet. Absorbing more through our skin is not good for our health over all.
Liquid soap may be slightly better for the simple fact that it is made of potassium hydroxide. Although it is still very caustic and very alkaline, our bodies can take higher doses of potassium than sodium. Also, most people don't meet their daily dose of potassium, so you may be a little safer there.
The pH of potassium hydroxide is very high as it is a form of lye as well. The resulting soap is similar to that of bar soap, so again it is raising the pH of your skin and leaving you exposed to harmful bacteria and fungus.
What Do You Use to Clean Your Skin
Facial Clensers are a Better Choice
To help maintain the skin's fatty protective layer, use a cleanser with a pH level similar to that of the skin itself. The pH level of many skin cleansers is slightly higher than 5.5, so the cleanser can break down dirt and oil on your skin.
Cleansers with lower pH levels leave your skin in tact instead of breaking down the acid mantle and the fatty tissue.
Helpful Hints to Avoid Skin Infections and Skin Fungus
Turn Down the Temperature:
Very hot water can also aid in stripping away your skins protective layer. That is what the tight feeling you get sometimes after washing your face is. Keeping your skin's natural oils is very important. You should use moderately warm water in your showers and baths to help avoid stripping your acid mantle.
Don't Over Exfoliate
While removing the dead skin cells and the plaque build up is necessary, try not to be too harsh with your skin. Being too aggressive with exfoliation strips your skin of its protective layer.
One of my favorite gentle exfoliates is rolled oats. Simply grind them up in a food processor or coffee grinder and gently scrub your skin.
Test pH Levels Yourself
Beauty products -- whether they're skin cleansers or shampoos -- don't often list their exact pH levels on the label, even when they claim to be pH-balanced. If you're curious about a product's pH, you can use litmus paper to test if a liquid is an acid, neutral or base. Even if the product label offers the pH which most wont, a litmus test will let you know for sure.
You Can Win the War of Skin Infections and Skin Fungus
Your skin flora is an important part of your skins health. By keeping the mutualistic bacteria happy you can prevent many skin infections and skin fungus. Keeping your skin's pH balanced will give your troops the advantage they need to keep your skin healthy.